-
dhiraj@gjpharmaceutical.com -
08048771781
Pharmaceutical Tablets
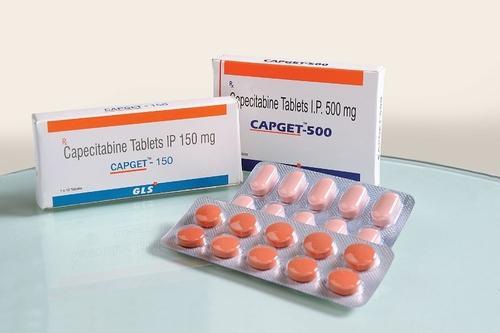
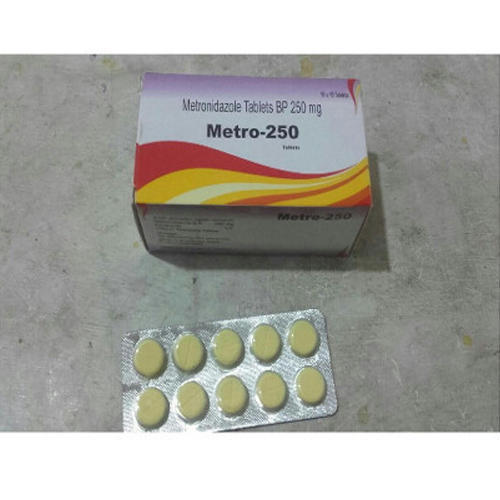
Leading Manufacturers, Exporters and Wholesaler of Capecitabine Tablets, Daclatasvir Dihydrochloride Tablets, Metronidazole Tablets and Nitroglycerine Sublingual Tablets from Thane.
| Business Type | Manufacturer, Exporter, Supplier |
| Packaging Size | Blister of 10 Tablets |
| Best Before | 24 months from Manufacture date |
| Brand Name | GJ |
| Strength | 500 mg |
| Form Of Medicine | Tablets |
| Storage | Store in cool and dry place |
Preferred Buyer From
| Location | All Countries Except India |
What Capecitabine Is Used For:
- Colon or rectal cancer
- Metastatic breast cancer
- Esophageal, gastric, hepatobiliary, neuroendocrine, pancreatic, ovarian, fallopian tube, peritoneal or unknown primary cancers (off-label use)
Note: If a drug has been approved for one use, physicians may elect to use this same drug for other problems if they believe it may be helpful.
How Capecitabine Is Given:
- Taken as a pill by mouth.
- Take after food (within 30 minutes of a meal) with water. (Usually taken in a divided dose 12 hours apart).
- Tablets come in 2 sizes; 150mg and 500mg.
- Do not crush, chew or dissolve tablets.
- If you miss a dose, skip the missed dose and go back to your normal time. Do not take 2 doses at the same time or extra doses.
The amount of capecitabine that you will receive depends on many factors, including your height and weight, your general health or other health problems, and the type of cancer or condition being treated. Your doctor will determine your dose and schedule.
Side Effects:
Important things to remember about the side effects of capecitabine:
- Most people will not experience all of the capecitabine side effects listed.
- Capecitabine side effects are often predictable in terms of their onset, duration, and severity.
- Capecitabine side effects will improve after therapy is complete.
- Capecitabine side effects may be quite manageable. There are many options to minimize or prevent the side effects of capecitabine.
Precautions:
- Before starting capecitabine treatment, make sure you tell your doctor about any other medications you are taking (including prescription, over-the-counter, vitamins, herbal remedies, etc.). Do not take aspirin, products containing aspirin unless your doctor specifically permits this.
- Avoid use of antacids within 2 hours of taking capecitabine.
- If you are on warfarin (Coumadin®) as a blood-thinner, adjustments may need to be made to your dose based on blood work.
- Capecitabine may be inadvisable if you have had a hypersensitivity (allergic) reaction to fluorouracil.
- Do not receive any kind of immunization or vaccination without your doctor's approval while taking capecitabine.
- Inform your health care professional if you are pregnant or may be pregnant prior to starting this treatment. Pregnancy category D (capecitabine may be hazardous to the fetus. Women who are pregnant or become pregnant must be advised of the potential hazard to the fetus).
- For both men and women: Do not conceive a child (get pregnant) while taking capecitabine. Barrier methods of contraception, such as condoms, are recommended. Discuss with your doctor when you may safely become pregnant or conceive a child after therapy.
Self-Care Tips:
- Drink at least two to three quarts of fluid every 24 hours, unless you are instructed otherwise.
- You may be at risk of infection so try to avoid crowds or people with colds, and report fever or any other signs of infection immediately to you health care provider.
- Wash your hands often.
- To help treat/prevent mouth sores, use a soft toothbrush, and rinse three times a day with 1/2 to 1 teaspoon of baking soda and/or 1/2 to 1 teaspoon of salt mixed with 8 ounces of water.
- Use an electric razor and a soft toothbrush to minimize bleeding.
- Avoid contact sports or activities that could cause injury.
- To reduce nausea, take anti-nausea medications as prescribed by your doctor, and eat small, frequent meals.
- Prevention of hand-foot syndrome. Modification of normal activities of daily living to reduce friction and heat exposure to hands and feet, as much as possible during treatment with capecitabine. (for more information see - Managing side effects: hand foot syndrome).
- Keeps palms of hands and soles of feet moist using emollients such as Aveeno®, Udder cream, Lubriderm® or Bag Balm®.
- Follow regimen of anti-diarrhea medication as prescribed by your health care professional.
- Eat foods that may help reduce diarrhea (see managing side effects - diarrhea).
- Avoid sun exposure. Wear SPF 30 (or higher) sunblock and protective clothing.
- You may experience drowsiness or dizziness; avoid driving or engaging in tasks that require alertness until your response to the drug is known.
- In general, drinking alcoholic beverages should be kept to a minimum or avoided completely. You should discuss this with your doctor.
- Get plenty of rest.
- Maintain good nutrition.
- If you experience symptoms or side effects, be sure to discuss them with your health care team. They can prescribe medications and/or offer other suggestions that are effective in managing such problems.
| Business Type | Manufacturer, Exporter, Supplier |
| Dose | 60mg |
| Pack Size | Single Vial |
| Brand Name | GJ |
| Packaging Size | 10 tablets per strip |
| Storage | Store in cool and dry place |
| Best Before | 24 months from Manufacture date |
Preferred Buyer From
| Location | All Countries Except India |
- Sustained virologic response (SVR12) rates are reduced in HCV genotype 3-infected patients with cirrhosis receiving DAKLINZA in combination with sofosbuvir for 12 weeks
NS5A Resistance Testing In HCV Genotype 1a-Infected Patients With Cirrhosis Consider screening for the presence of NS5A polymorphisms at amino acid positions M28, Q30, L31, and Y93 in patients with cirrhosis who are infected with HCV genotype 1a prior to the initiation of treatment with DAKLINZA and sofosbuvir with or without ribavirin
Tablets:
- 60 mg: 60 mg of daclatasvir (equivalent to 66 mg daclatasvir dihydrochloride), light green, biconvex, pentagonal, and debossed with “BMS” on one side and “215” on the other side.
- 30 mg: 30 mg of daclatasvir (equivalent to 33 mg daclatasvir dihydrochloride), green, biconvex, pentagonal, and debossed with “BMS” on one side and “213” on the other side.
- 90 mg: 90 mg of daclatasvir (equivalent to 99 mg daclatasvir dihydrochloride), light green, biconvex, round, and embossed with “BMS” on one side and “011” on the other side.
- If DAKLINZA and sofosbuvir are administered with ribavirin, refer to the prescribing information for ribavirin regarding ribavirin-associated adverse reactions.
- The following serious adverse reaction is described below and elsewhere in the labeling:
- Serious Symptomatic Bradycardia When Coadministered with Sofosbuvir and Amiodarone
| Business Type | Manufacturer, Exporter, Supplier |
| Best Before | 3 Years from the Date of Manufacturing |
| Available Strength | 200 mg |
| Medicine Type | Allopathic |
| Form | Tablets |
| Storage | Cool & Dry Place |
| Dosage | As per doctor's prescription |
| Strength | 200 & 400 mg |
| Pack Size | 10 Tablets |
Preferred Buyer From
| Location | All Countries Except India |
| Business Type | Manufacturer, Exporter, Supplier |
| Best Before | 24 months from Manufacture date |
| Form Of Medicine | Tablets |
| Type of Medicine | Allopathic |
| Storage | Store in cool and dry place |
| Dosage Strength | 8 mg |
| Doses | As per doctors prescription |
Preferred Buyer From
| Location | All Countries Except India |